Stem Cell Hair Treatment: Evidence, Results & Cost (2026 Guide)
Stem cell hair treatment uses a patient's own stem cells (typically drawn from fat tissue (ADSCs)) injected into the scalp to stimulate miniaturized hair follicles. Small peer-reviewed studies in 2020–2024 show a 20–30% average increase in hair density over six months, but no stem cell hair therapy is FDA-approved as of 2026, results vary between patients, and effects are not permanent.
Table of Contents
Dr. Terziler Clinic does not offer stem cell hair therapy. Our focus is health first, beauty second: a root-driven approach that extends across a lifetime, including our Reset Program, which treats hair and skin as reflections of deeper genetic and metabolic wellbeing. This guide is educational and evidence-first.
Stem Cell Hair Treatment in 60 Seconds
- Stem cell hair treatment is a regenerative, injection-based procedure, not a transplant.
- Evidence in 2026 shows a 20–30% density increase over 6 months in small trials, but is not yet FDA-approved.
- It works for early–moderate thinning with existing follicles; it does not regrow hair in fully bald zones.
- Results last 12–18 months on average; maintenance sessions are required.
- US costs range $3,000–$15,000; Turkey costs $1,500–$6,000.
- FUE/DHI hair transplantation remains the gold standard for permanent density in bald zones.
- Dr. Terziler Clinic does not offer stem cell hair therapy, we prioritize procedures with long-term outcome data.

MEDICALLY REVIEWED BY
Dr. Servet TerzilerAACI-accredited surgeon | Founder & President, TUSATDER | Creator, Picasso Robotic DHI | European Property Awards — Best Hair Transplant Clinic in Europe
Last reviewed April 17, 2026
What Is Stem Cell Hair Treatment?
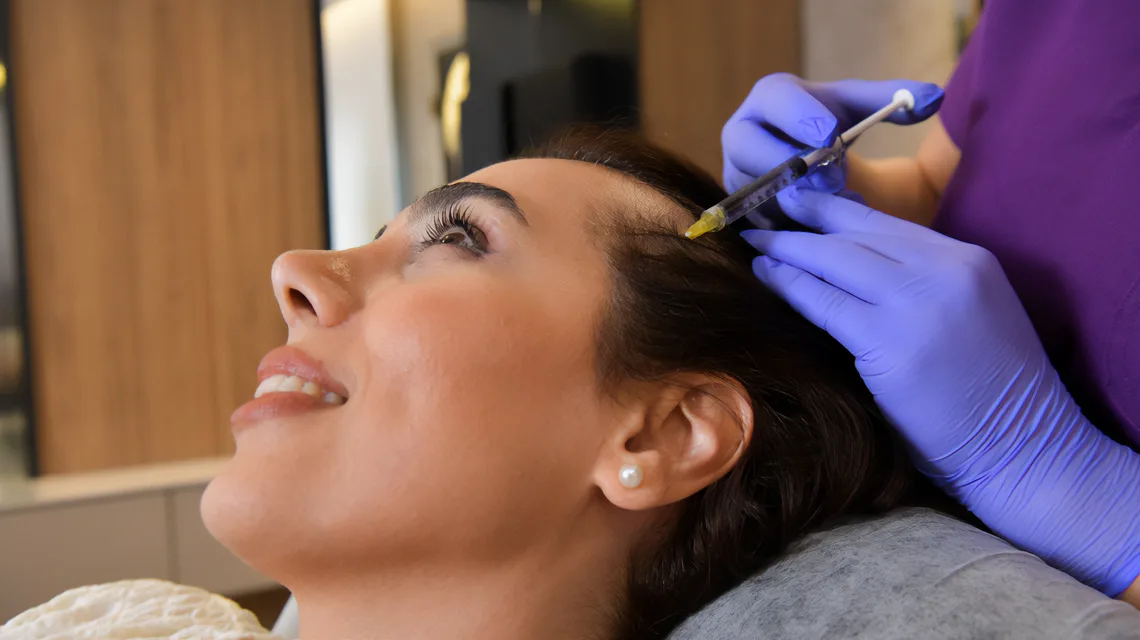
Stem cell hair treatment is a regenerative procedure in which mesenchymal stem cells (most commonly adipose-derived stem cells (ADSCs) harvested from a patient's own fat) are processed and injected into the scalp to reactivate miniaturized hair follicles. The goal is to increase hair thickness and density in areas where follicles are still present but producing thinner, weaker hairs. It is not a transplant procedure and does not move follicles from one part of the scalp to another.
The field brings together three ideas: (a) hair follicles contain their own reservoir of stem cells in the bulge region; (b) injected mesenchymal stem cells release growth factors (including VEGF, IGF-1, and PDGF) that signal follicles to return to the anagen (growth) phase; and (c) in most modern protocols, the cellular product is prepared the same day as the scalp injection, often from a small abdominal or flank fat harvest performed under local anesthesia.
Autologous vs Allogeneic Stem Cells
Virtually all legitimate 2026 clinical protocols use autologous stem cells: the patient's own cells. Allogeneic (donor-derived) products, including Wharton's jelly umbilical-cord preparations, are heavily restricted in the United States and regulated as drugs in the European Union. Autologous-only preparations are the only category generally considered within the practice of medicine rather than as investigational drug products.
How It Differs from PRP, Exosomes, and Hair Transplants
PRP (platelet-rich plasma) uses growth factors from a patient's own blood, not cells. Exosome therapy uses the vesicle-borne signaling molecules that stem cells secrete, without using the cells themselves. FUE/DHI hair transplantation physically relocates follicles from DHT-resistant donor zones; those transplanted follicles are permanent. Stem cell therapy is a fundamentally different from these hair treatments: it is a signal-and-stimulate intervention, not a relocation procedure.
How Does Stem Cell Hair Restoration Work?
A typical modern stem cell hair protocol involves harvesting 20–50 mL of the patient's own fat, isolating the stromal vascular fraction containing adipose-derived stem cells, and injecting the prepared product across the thinning scalp zone in one session lasting 2–3 hours. Growth factors secreted by the stem cells then act on existing follicles over the following weeks.
Step-by-step procedure:
- Consultation and mapping. Scalp trichoscopy, Norwood/Ludwig grading, blood workup (CBC, coagulation, HIV/HCV/HBV screening).
- Fat harvest. Local anesthesia, 3–5 mm cannula liposuction from abdomen or flank, 20–50 mL lipoaspirate.
- Cell isolation. Enzymatic digestion or mechanical disruption, centrifugation, isolation of stromal vascular fraction (SVF) containing ADSCs, viability check.
- Scalp preparation. Topical anesthesia, optional microneedling to increase penetration.
- Injection. Mesotherapy-style pattern across thinning areas, typically 3–6 mL total volume distributed over 100–200 injection points.
- Aftercare. No shampoo for 24 hours, no sun exposure for 72 hours, no exercise for 48 hours.
Stem Cell Sources: ADSC, Bone Marrow, Wharton's jelly
Adipose-derived stem cells dominate because fat yields 100–1,000 times more mesenchymal stem cells per gram than bone marrow. Bone marrow aspiration (BMAC) is painful, low-yield, and now uncommon for hair indications. Wharton's jelly preparations are legally restricted in most jurisdictions and should be regarded with caution when offered outside a formal clinical trial.
Does Stem Cell Hair Treatment Actually Work?
Current evidence is cautiously positive for androgenic alopecia in early-to-moderate stages but remains preliminary. Across the most-cited peer-reviewed studies from 2020 to 2024, patients typically show a 20–30% increase in hair density measured by phototrichogram over six months, with response rates of 60–80%. None of these trials is large enough or long enough to support general clinical claims of efficacy, and all authors explicitly caution that stem cell hair therapy is not yet a replacement for established treatments.
- Anderi et al., 2018: autologous SVF injection in 20 androgenic alopecia patients, hair count increased 31% at 6 months.
- Tak et al., 2020: 29 patients treated with adipose-derived stem cell conditioned media, average 28.1% increase in hair count at 16 weeks (PubMed).
- Egger et al., 2020: systematic review in Plastic and Reconstructive Surgery of 9 trials including 207 patients, concluded benefit but called for RCTs (PubMed).
- NIH PMC review, 2020: "Advances in Stem Cell-Based Therapy for Hair Loss" (PMC7508456): confirms mechanism but notes absence of long-term follow-up and standardized protocols.
Realistic Expectations: Density, Coverage, Timeline of Stem Cell Hair Treatment
- Month 1: Minor shedding (telogen effluvium) possible, no visible improvement yet.
- Month 3: Early density improvement, less noticeable thinning in good light.
- Month 6: Peak density measurable by trichoscopy (typically +20–30% hair count).
- Month 12: Results begin to plateau; touch-up often recommended at 12–18 months.
Who is not a candidate for Stem Cell Hair Treatment?
Stem cell hair therapy is not suitable for fully bald zones, active scalp infections, uncontrolled diabetes, bleeding disorders, active autoimmune disease, current chemotherapy, or for scar-tissue regrowth expectations. End-stage Norwood 6 and 7 patterns respond poorly because the follicular unit itself has disappeared.
Stem Cell Hair Treatment vs FUE vs DHI Hair Transplant
FUE and DHI hair transplants and stem cell hair therapy solve different problems and are not direct substitutes. FUE physically moves DHT-resistant follicles from the donor area to bald zones using micro-punches; the result is permanent, well documented across decades, and covered by clear clinical guidelines. DHI uses the same DHT-resistant follicles but implants them with a Choi pen without pre-opened channels, allowing denser packing and more control over hairline angle and direction. Stem cell hair therapy is a regenerative adjunct that only works where follicles still exist; as a 2026 standalone solution, the research base does not yet justify replacing FUE or DHI with it.
| Criterion | Stem Cell Hair Therapy | FUE Hair Transplant | DHI Hair Transplant |
|---|---|---|---|
| Mechanism | Stimulates existing follicles with growth factors / regenerative cells | Relocates DHT-resistant follicles via micro-punch extraction + pre-made channels | Relocates DHT-resistant follicles via Choi implanter pen, no pre-made channels |
| Best for | Early–moderate thinning where follicles are still alive | Moderate–advanced baldness, large session sizes, crown coverage | Hairline design, high-density front zones, unshaven or partially shaved procedures |
| Permanence | 12–18 months, then touch-up | Lifetime for transplanted grafts | Lifetime for transplanted grafts |
| Evidence base | Promising but no FDA clearance; limited long-term RCTs | Decades of peer-reviewed clinical evidence | Variant of FUE; well-documented, strong real-world evidence |
| Typical cost (USA) | $3,000–$15,000 per session | $4,000–$15,000 total | $7,000–$20,000 total |
| Typical cost (Turkey) | $1,500–$6,000 | $1,800–$4,500 | $2,500–$8,000 |
| Downtime | 1–3 days | 7–10 days | 7–10 days |
| Visible result timing | 3–6 months | 8–12 months | 8–12 months |
| Shaving required | No | Usually full or partial shave | Often unshaven or partial shave possible |
| Session length | 1–2 hours | 6–8 hours | 8–10 hours |
For thorough reading on DHI specifically, see our DHI hair transplant and Robotic DHI guide.
Stem Cell Hair Treatment: Before and After Timeline
Published before-and-after series for stem cell hair therapy typically show modest but visible density gains between months 3 and 6, with peak effect around month 6 and stabilization by month 12. Unlike FUE, changes are not dramatic single-event transformations, they are incremental improvements in existing hair.
- Month 0 (baseline): Standardized photography at 10 cm, full-scalp trichoscopy, hair count per cm².
- Month 1: Possible mild shedding, no visible improvement.
- Month 3: Early density improvement visible in photographs under consistent lighting.
- Month 6: Peak measurable improvement, typically +20–30% density in responders.
- Month 12: Stabilization; maintenance session considered at 12–18 months.
We recommend that any patient evaluating before-and-after images for stem cell hair therapy ask for (a) standardized lighting, (b) phototrichogram data rather than photos alone, and (c) full disclosure of any adjunctive treatments (PRP, finasteride, minoxidil) used in the same period.
Side Effects & Safety of Stem Cell Hair Treatment
In published 2020–2024 clinical literature, autologous stem cell hair therapy has a favorable short-term safety profile with no serious adverse events reported, but long-term (>2 year) safety data remain limited. Common side effects are local and resolve within a week; rare but serious risks include infection, inflammation, and (with donor-derived cells) immune reaction.
Common side effects (minor)
- Scalp tenderness and mild pain for 24–72 hours (reported in ~60% of patients)
- Swelling or bruising at injection sites (10–20% of patients)
- Temporary itching at the injected zone
- Small post-harvest bruising at the fat-donor area
- Brief telogen effluvium (shedding) in the first 3–4 weeks
Rare risks & contraindications
- Infection at the injection or harvest site (reported <1% in clinical series)
- Localized fibrosis or granuloma formation (very rare with autologous cells)
- Immune reaction (only with allogeneic / donor-derived cells)
- Failure to respond (10–40% of patients across studies)
Regulatory status (2026)
- United States - FDA: no stem cell hair therapy is FDA-approved; the agency has issued multiple warnings against clinics selling "stem cell cures" (FDA consumer update).
- Turkey - Ministry of Health: autologous stem cell procedures are permitted within licensed medical centers under the cell therapy directive; allogeneic products require clinical trial authorization.
- European Union - EMA: stem cell preparations are regulated as advanced therapy medicinal products (ATMPs).
How Much Does Stem Cell Hair Treatment Cost?
Global price ranges in 2026 run from $1,500 in Turkey to $15,000 per session in major US cities. Price variance reflects cell-product complexity (SVF vs culture-expanded), session count, adjunctive therapies, and regulatory overhead.
| Country | Low | High | Typical sessions |
|---|---|---|---|
| United States | $3,000 | $15,000 | 1–3 |
| Mexico | $1,800 | $6,000 | 1–3 |
| United Kingdom | £2,500 | £8,000 | 1–3 |
| Germany | €3,000 | €9,000 | 1–3 |
| Turkey | $1,500 | $6,000 | 1–3 |
What influences the price
- Cell source: autologous SVF (same-day) vs culture-expanded cells (multi-week lab work), the latter roughly doubles the cost.
- Number of sessions: most published protocols use 1–3 sessions spaced 4–8 weeks apart.
- Adjunctive therapies: PRP, exosomes, or microneedling combined with the stem cell session add $300–$1,500.
- Clinic overhead: US regulatory compliance, lab processing, and physician fees drive the USA premium.
For hair transplant cost comparisons, see our detailed hair transplant cost guide (Turkey).
Does Dr. Terziler's Clinic Offer Stem Cell Hair Treatment?
No. Dr. Terziler Clinic does not currently offer stem cell hair therapy as a standalone treatment. We prioritize procedures with decades of long-term outcome data.
Related Emerging Research: Hair Cloning, Exosomes & What's Next
Hair cloning and exosome therapy are the two most-discussed adjacent fields in 2026 regenerative hair medicine. Both are distinct from stem cell hair therapy as it is practiced today.
Hair cloning (follicular neogenesis)
Hair cloning proposes to multiply a patient's own follicles in a laboratory and then transplant the cultured follicles. Two leading pipelines (dNovo (San Francisco) and Stemson Therapeutics) are in preclinical and early clinical stages as of 2026. No hair cloning product is commercially available; any clinic claiming to sell "hair cloning" today is using a different procedure under a misleading label.
Exosome Hair Treatment
Exosomes are small vesicles secreted by stem cells that carry growth-factor and RNA cargo. Exosome therapy uses the cargo without the cells. In 2024–2025 several clinical series reported comparable density gains to ADSC therapy with fewer procedural steps. Exosome products intended for injection remain under FDA scrutiny in the US; availability varies.